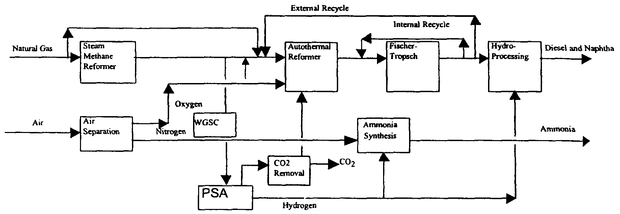
 This is how ammonia is produced.
This is how ammonia is produced.
Resource Extraction:
As previously said in the manufacturing process, the raw materials that creates sodium bicarbonate are limestone, ammonia, and sodium chloride. These resources come from different places and go through alot of operations to create sodium bicarbonate.
1. Limestone also known as calcium carbonate (CaCO3) is extracted from the ground. For example: Calcium carbonate can be found in the Earth's crust as well as marble, oyster shells, pearls, chalk, and etc,. This compound can be found by mining natural sources like chalk, marble quarries, and limestone. Calcium Carbonate can be produced by combining calcium chloride (CaCl2) with sodium carbonate (Na2CO3) together. The calcium carbonate that is formed in the reaction precipitates out of the solution. The calcium carbonate can be removed from the solution through filtration (Calcium Carbonate, 2006).
Limestone is a sedimentary rock and is found in areas that contains an abundance of sedimentary rocks. An example of one place that has sedimentary rocks would be Niagara Falls (personal communication, February 14, 2014). According to Niagara Parks, these falls have a limestone bed resting underneath it's waters.
2. Ammonia is formed during the decay of dead organisms. Ammonia was produced commercially, by the end of the 19th century, by the cyanamide process. Calcium carbide (CaC2), nitrogen gas (N2), and steam were combined together to produce ammonia.
In the early 1900s, a German chemist, Fritz Haber had created a method to make ammonia (NH3) directly from its elements, nitrogen and hydrogen. These two gases were combined with each other at high temperature and pressure over a catalyst. As years passed by, Fritz Haber's process was refined and improved by, a German chemical engineer, Carl Bosch. The Haber-Bosch process remains as the most common form of producing ammonia (Ammonia, 2006).
3. The simpliest method known to obtain sodium chloride (NaCl) is the evaporation of the seawater. In this procedure, Seawater is collected in large, shallow ponds. This way, the seawater has a faster rate of evaporating. The salts that are dissolved in the water harden and rest on the bottom of the ponds. Once they are hardened, they are scraped off from the bottom of the floor. The individual compounds that are present, including sodium chloride, are separated from each other (Sodium Chloride, 2006).
As previously said in the manufacturing process, the raw materials that creates sodium bicarbonate are limestone, ammonia, and sodium chloride. These resources come from different places and go through alot of operations to create sodium bicarbonate.
1. Limestone also known as calcium carbonate (CaCO3) is extracted from the ground. For example: Calcium carbonate can be found in the Earth's crust as well as marble, oyster shells, pearls, chalk, and etc,. This compound can be found by mining natural sources like chalk, marble quarries, and limestone. Calcium Carbonate can be produced by combining calcium chloride (CaCl2) with sodium carbonate (Na2CO3) together. The calcium carbonate that is formed in the reaction precipitates out of the solution. The calcium carbonate can be removed from the solution through filtration (Calcium Carbonate, 2006).
Limestone is a sedimentary rock and is found in areas that contains an abundance of sedimentary rocks. An example of one place that has sedimentary rocks would be Niagara Falls (personal communication, February 14, 2014). According to Niagara Parks, these falls have a limestone bed resting underneath it's waters.
2. Ammonia is formed during the decay of dead organisms. Ammonia was produced commercially, by the end of the 19th century, by the cyanamide process. Calcium carbide (CaC2), nitrogen gas (N2), and steam were combined together to produce ammonia.
In the early 1900s, a German chemist, Fritz Haber had created a method to make ammonia (NH3) directly from its elements, nitrogen and hydrogen. These two gases were combined with each other at high temperature and pressure over a catalyst. As years passed by, Fritz Haber's process was refined and improved by, a German chemical engineer, Carl Bosch. The Haber-Bosch process remains as the most common form of producing ammonia (Ammonia, 2006).
3. The simpliest method known to obtain sodium chloride (NaCl) is the evaporation of the seawater. In this procedure, Seawater is collected in large, shallow ponds. This way, the seawater has a faster rate of evaporating. The salts that are dissolved in the water harden and rest on the bottom of the ponds. Once they are hardened, they are scraped off from the bottom of the floor. The individual compounds that are present, including sodium chloride, are separated from each other (Sodium Chloride, 2006).
