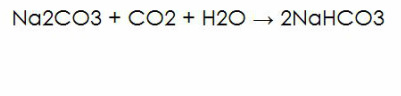 This is one of the formulas of creating sodium bicarbonate.
This is one of the formulas of creating sodium bicarbonate.
How is it made?
One of the ways of creating sodium bicarbonate is passing carbon dioxide gas through an aqueous solution of sodium carbonate. Since the bicarbonate is less soluble than the carbonate, a precipitate is formed and can be removed by filtration. Sodium bicarbonate can also be obtained as a byproduct of the solvay process. (Sodium Bicarbonate, 2006)
Raw materials:
According to the Solvay process, sodium bicarbonate is created from a precipitate from a solution and removed by filtration. The required raw materials that are needed in this process are:
Materials Process:
Around the 1850s, A belgian chemist, Ernest Solvay, created a procedure called the " Solvay Process".
Step 1: Sodium chloride is combined with carbon dioxide and ammonia.
Step 2: The results from the combination forms sodium bicarbonate and ammonium bicarbonate.
Step 3: The sodium bicarbonate is heated to obtain the sodium carbonate.
Step 4: Sodium bicarbonate can be obtained by removing the last step. (Sodium Bicarbonate, 2006)
One of the ways of creating sodium bicarbonate is passing carbon dioxide gas through an aqueous solution of sodium carbonate. Since the bicarbonate is less soluble than the carbonate, a precipitate is formed and can be removed by filtration. Sodium bicarbonate can also be obtained as a byproduct of the solvay process. (Sodium Bicarbonate, 2006)
Raw materials:
According to the Solvay process, sodium bicarbonate is created from a precipitate from a solution and removed by filtration. The required raw materials that are needed in this process are:
- Lime stone (Caco3)
- Sodium chloride (NaCl)
- Ammonia (NH3)
(Tutorvista,2013)
Materials Process:
Around the 1850s, A belgian chemist, Ernest Solvay, created a procedure called the " Solvay Process".
Step 1: Sodium chloride is combined with carbon dioxide and ammonia.
Step 2: The results from the combination forms sodium bicarbonate and ammonium bicarbonate.
Step 3: The sodium bicarbonate is heated to obtain the sodium carbonate.
Step 4: Sodium bicarbonate can be obtained by removing the last step. (Sodium Bicarbonate, 2006)
